One of the most impactful changes coming up in the regulatory publishing landscape is the global implementation of eCTD version 4.0. Here, one of Calyx’s Senior RIM consultants, Diarmuid Waide, reviews the features of eCTD v4.0 and the latest timelines.
eCTD v4.0 Features
Context of Use Keywords
This new feature enables users to perform new life cycle operations on documents such as one-to-many and many-to-one. This means easier manipulation of document metadata to fix inconsistencies. Additionally, documents will be grouped under one common theme and prioritized within the dossier.
One Submission Unit
Previously eCTD featured a M1 specific XML and a M2-M5 XML. Under eCTD Version 4.0 there will be one XML that covers all modules. This decreases complexity for the end user.
Document Re-use
Once submitted, a document may be reused by referring to its unique identifier (ID). This will be helpful to users who submit grouped submissions relating to similar products. Document reuse will speed up publishing times since the end user will be able to send a document just once and use the ID to reference the document in other submissions.
Controlled Vocabularies (CVs)
Controlled Vocabularies are a list of pre-defined terms to be used in submissions. This will aid the agencies in defining what they want to see in the metadata and can be easily updated without the need for changes in tools. This in turn reduces the time it takes to adapt to new regulatory landscapes. User defined CVs can be updated and replaced in subsequent submissions with ease.
Document Metadata
There also has been updates to document metadata to help the user define the content of the submissions such as datasets. Increasing visibility of such documents to the reviewer can allow for efficiency gains during the review process.
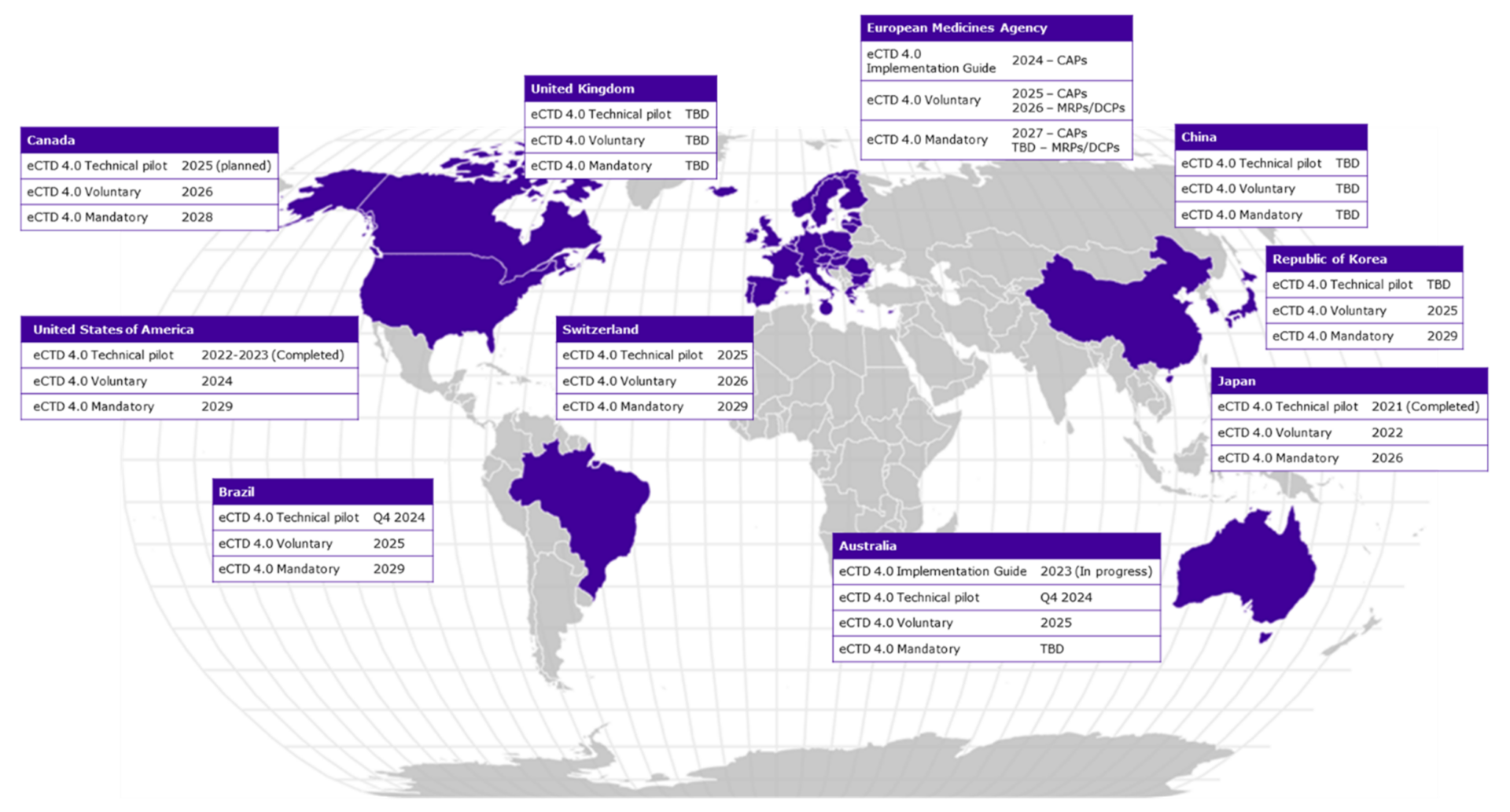
Timelines for Regional Implementation
With the advantages of eCTD version 4.0 clear, it’s time to look at when you can expect this update to come to the major markets:
2023 was a busy year for eCTD v4.0. The FDA pilot, in which Calyx was proud to support a market-leading pharmaceutical company, was a key step towards implementation in the USA. In 2024, we expect to see further technical pilots in Australia and Brazil, although these dates have changed in the past and remain subject to change. We also expect to see further Implementation Guides issued by the EMA and the TGA and for the FDA to begin accepting voluntary submissions in eCTD v4.0 format.
We’ll continue to closely monitor the progress of eCTD v4.0 implementation and work with our clients on eCTD 4.0 implementation readiness. With decades of regulatory and user experience, Calyx RIM can always be relied upon to support you in continuing to meet regulatory requirements worldwide.
Learn how Calyx RIM helps you maintain compliance with all regulatory requirements.